 
The Group employs more than 6,000 people. The Group’s Research and Development centre is based in Parma and works alongside 5 other important research and development centres in France, the U.S., Canada, the UK and Sweden to promote its pre-clinical, clinical and regulatory programmes. It conducts research and develops and markets innovative drugs for respiratory treatment, Special Care and rare diseases. 4-6īased in Parma, Italy, Chiesi Farmaceutici is an international, research-focussed group with 85 years’ experience in the pharmaceutical sector operating in 30 countries. 1,2 The CHMP recommendation and the European Commission decision are based on the efficacy and safety data of four clinical studies involving close to 3000 patients. In patients with uncontrolled asthma, Chiesi triple therapy has been shown to reduce exacerbations and improve lung function in comparison to ICS/LABA. 8 According to GINA, people with uncontrolled asthma have poor symptom control and/or frequent exacerbations requiring oral corticosteroids or experience serious exacerbations requiring hospitalisation. 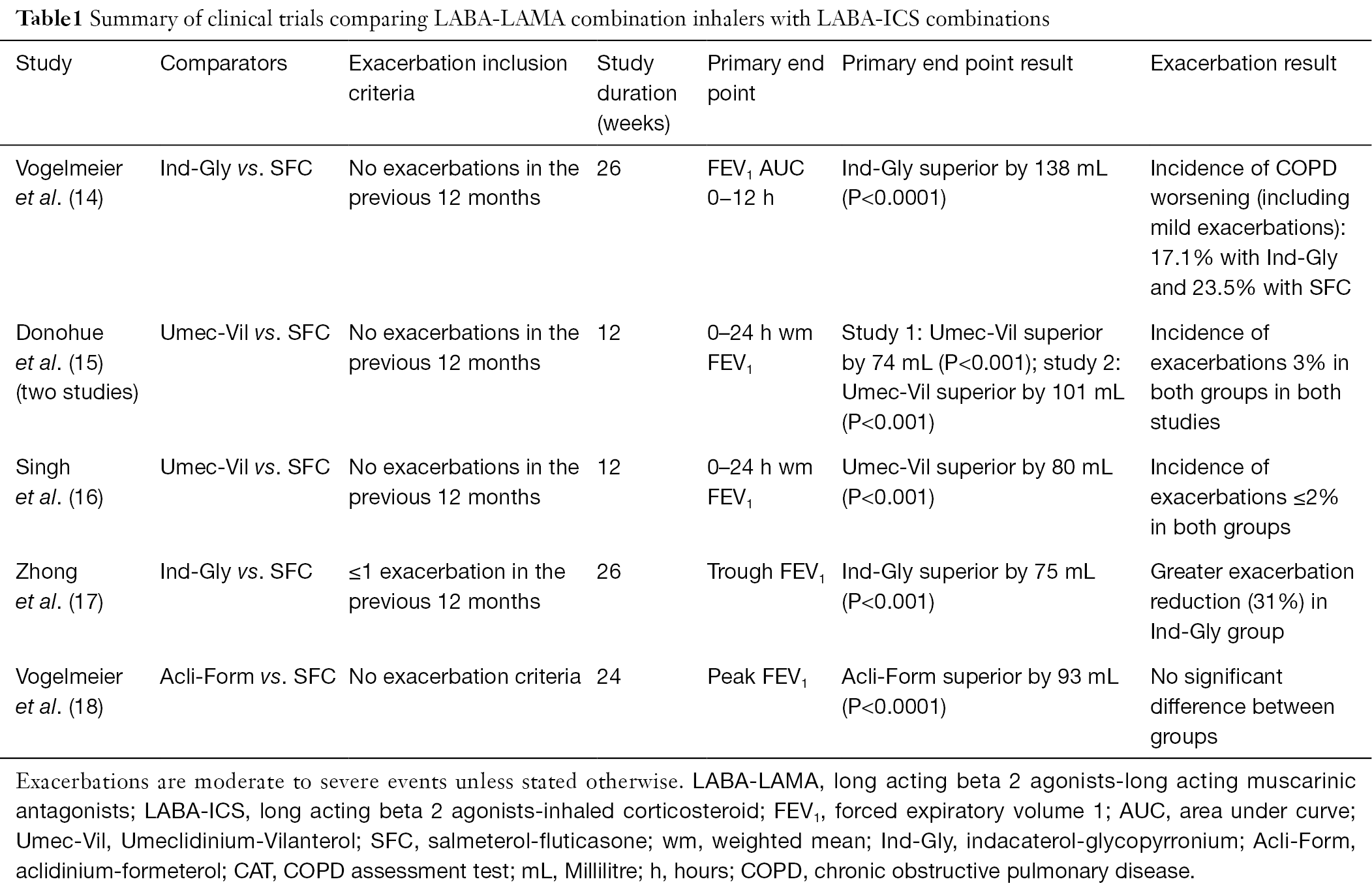
The European Commission’ approval brings us one step closer to providing uncontrolled adult asthma patients with a new treatment option for their care, reducing exacerbations and potentially simplifying the use for patients, thanks to a single inhaler triple therapy.”Īsthma is a chronic inflammatory disease affecting over 339 million people worldwide. “ Chiesi is committed to developing and delivering alternatives to aid patients in the management of their respiratory conditions and treatment. “This is the first step towards taking our triple therapy for moderate to severe COPD patients and providing the same treatment option for appropriate asthma patients,” said Alessandro Chiesi, Chiesi Group Chief Commercial Officer. 7 The inhaler also contains a dose counter that allows patients to track and manage their treatment. 
1,2 For asthma, this is the first triple, extrafine formulation, combination inhaler with the ability to reach the whole bronchial tree, including both the large and the small airways. 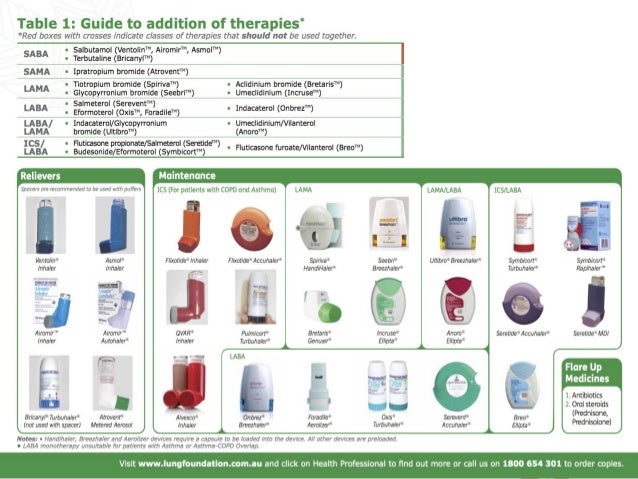
3Ĭhiesi’s triple therapy is a combination of an ICS/LABA/LAMA that contains beclometasone dipropionate (ICS), formoterol fumarate (LABA) and glycopyrronium (LAMA). 1,2 This follows the 2017 approval of this therapeutic option for the treatment of adult patients with moderate to severe chronic obstructive pulmonary disease (COPD) who are not adequately treated by a combination of an ICS/LABA, or a combination of a LABA/LAMA (long-acting muscarinic antagonist). Parma, Italy, Febru– Chiesi, an international research-focused healthcare group (Chiesi Group), announces that the European Commission has granted the marketing authorisation for Trimbow (beclometasone/formoterol/glycopyrronium), an extrafine formulation triple fixed combination therapy in a single inhaler, as a maintenance treatment for adult asthma patients not adequately controlled with a maintenance combination of a long-acting beta2-agonist (LABA) and medium dose of inhaled corticosteroid (ICS), who experienced one or more asthma exacerbations in the previous year. The Marketing Authorisation approval is based on Committee for Medicinal Products for Human Uses’ positive opinion and European Commission approval on data obtained in four clinical studies involving close to 3000 patients.1,2 This follows the 2017 approval of this therapeutic option for the treatment of appropriate moderate to severe chronic obstructive pulmonary disease (COPD) patients. Trimbow is the first extrafine formulation fixed triple combination therapy in a single inhaler to be approved for use in appropriate asthma patients. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
